Below is our recent interview with Kim Kundert, VP of Clinical Operations at VirTrial:
Q: Could you provide our readers with a brief introduction to VirTrial?
A: VirTrial is a telehealth platform provider in the clinical research space. VirTrial’s founder, Mark Hanley is an industry veteran who most recently served as the CEO of Radiant Research/Synexus, the country’s largest wholly-owned network of clinical trial sites. Based on his 11+ years of site knowledge conducting more than 10,000 trials and enrolling hundreds of thousands of patients, Hanley recognized the need to find a better way to engage and retain patients in trials.
In 2018, Hanley founded VirTrial with a goal of transforming telehealth in the clinical research industry. To ensure a reliable solution, he acquired a stable, award-winning virtual care platform and customized it for clinical trial use. His vision is to replace 25-40 percent of standard clinical trial visits with virtual visits to create hybrid studies.
Q: How exactly does it work?
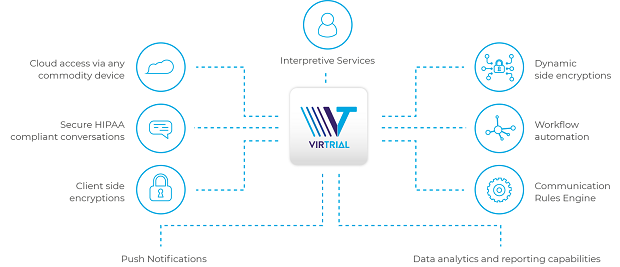
A: VirTrial is built on a proven telehealth platform that won the 2018 Medtech breakthrough award for virtual care innovation. The platform has been in use for the past eight years and is used at over 1700 hospitals, conducting 100,000 virtual visits per day. The app has been customized for clinical research use, is HIPAA-compliant and fully secure. Using this proven technology, and based on sponsor protocol, sites can offer virtual visits to patients for some of their visits.
Virtual visits are an incredibly useful solution to incorporate into traditional clinical trials. They are ideal for visits that require things like a simple compliance check which can safely and easily be accomplished remotely. By enabling patients to use their mobile devices to communicate via phone, email, or video, they can more easily comply with study requirements and benefit from their participation in the program.
Q: Can you give us insights into your features?
A: The VirTrial leadership team has spent their entire careers in sites and have conducted over 10,000 trials. That experience has led VirTrial to take a unique approach to providing virtual visits that make it easier on both patients and research staff.
● Hybrid approach – VirTrial supports clinical trials that require a combination of in-clinic and virtual visits, unlike other providers that promote ‘site-less’ trials.
● Research site agnostic – VirTrial is available to any clinical research site and provides training to PIs and site staff.
● Device agnostic – Patients can use personal mobile devices that they’re already comfortable using, including Apple, Android, smartphone, tablet, desktop. This eliminates the need to carry a second device, which other providers require.
● Up to 8-way calling – Unlike in-person visits, it is possible to add a family member, caregiver or another physician to the conversation simply by enabling the feature.
● Interpretation services – PIs can easily add an interpreter to a call when needed. VirTrial offers 20 languages via video and 183 via audio, all with medically certified interpreters.
Q: Why do we need virtual platforms for clinical trials?
A: VirTrial works in conjunction with traditional research sites to better enable patient engagement and retention in clinical trials. Continual in-person visits to a site can be a burden on patients and they often can’t continue through the course of an entire trial. By incorporating virtual trials into the mix and enabling patients to comply with the required visit schedule by conducting some visits virtually from their home or workplace makes it much easier for them to complete the trial and avoid delays.
Trials using only in-person visits often take longer than anticipated due to an average 30% drop out rate in standard phase III trials. The dropout rate can decrease to only 5%* in virtual trials. With the average cost of a dropped patient being $5000** and an average clinical trial including 1000 patients, the resulting savings ranges around $1.25M per trial.
Q: You’ve recently received acclaim as leader in life sciences solutions – what does it mean for the company?
A: VirTrial was recently recognized by CIOReview as one of the ‘20 most promising pharma & life sciences tech solution providers of 2018.’ The Innovative app also was selected as a finalist for the ‘Fierce Innovation Award – Life Sciences Edition 2018’ in the Digital Health Solutions Category, by FierceBiotech.
We’re honored for the recognition we have received so far and will continue our work to transform the use of telemedicine in the clinical research industry. Our patient management program combines video, text, and email for clinical trial sites to easily and conveniently address specific patient and/or study needs in a secure environment.
 Recommended: Meet Yooz – A Fast-Growing, Multiple Award-Winning SaaS Company
Recommended: Meet Yooz – A Fast-Growing, Multiple Award-Winning SaaS Company
Q: What are your plans for the future?
A: VirTrial recently became a Global Impact Partner (GIP) for the Society for Clinical Research Sites (SCRS) to further support the inclusion of research sites in the evolution of clinical trials. As part of our commitment to site sustainability, we will be offering VirTrial’s telemedicine platform to all SCRS member sites as part of their SCRS membership.
From a technology perspective, VirTrial plans to include the capability for participants to sign consent forms electronically, and is slated for API integration to wearables and other technology applications such as ePRO and edc. We also intend to enable sites to use our platform for purposes outside of trials, such as general patient communication.

 Recommended:
Recommended: 